Alphabetical

isoelectronic
[adjetivo]
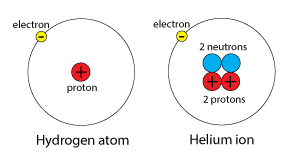
Different atomic species that have the same electronic configurations. For example, hydrogen atoms and helium ions (He+1) are isoelectronic because the two species each exist with a single valence electron.
Aparece en los módulos:
Ingresa o Registro
Para disfrutar de una experiencia sin publicidad y acceder a Visionlearning Classroom, inicie sesión o regístrese.
