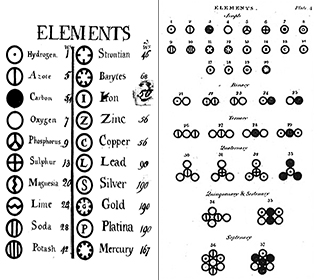
Early Ideas about Matter: From Democritus to Dalton
by Anthony Carpi, Ph.D.
Listen to this reading
00:00Did you know that some ancient Greeks believed that all matter was made up of four substances: fire, air, water, and earth? They believed that rabbits were soft because they had more water than earth. Although this idea seems silly now, it contains a fundamental principle of atomic theory: that matter is made up of a small number of fundamental elements.
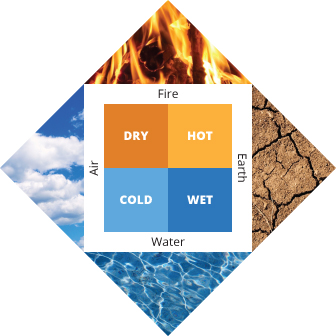
Early humans easily distinguished between materials that were used for making clothes, those that could be shaped into tools, or those that were good to eat. Then they gave these things the names, such as "fur," "stone," or "rabbit." However, these people did not have our current understanding of the substances that made up those objects. Empedocles, a Greek philosopher and scientist who lived on the south coast of Sicily between 492 BCE and 432 BCE, proposed one of the first theories that attempted to describe the things around us. Empedocles argued that all matter was composed of four elements: fire, air, water, and earth. The ratio of these four elements affected the properties of the matter. Stone was thought to contain a high amount of earth, while a rabbit was thought to have a higher ratio of both water and fire, thus making it soft and giving it life.

Empedocles's theory was quite popular, but it had a number of problems. For example, regardless of how many times you break a stone in half, the pieces never resemble any of the core elements of fire, air, water, or earth. Despite these problems, Empedocles's theory was an important development in scientific thinking because it was among the first to suggest that some substances that looked like pure materials, like stone, were actually made up of a combination of different "elements."
The atom is proposed
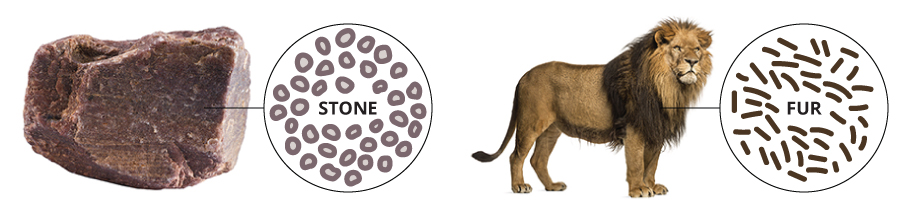
A few decades after Empedocles, Democritus (460 BCE - 370 BCE), who was also Greek, developed a new theory of matter that attempted to overcome the problems of his predecessor. Democritus's ideas were based on reasoning rather than science, and drew on the teachings of two Greek philosophers who came before him: Leucippus and Anaxagoras. Democritus knew that if you took a stone and cut it in half, each half had the same properties as the original stone. He reasoned that if you continued to cut the stone into smaller and smaller pieces, at some point you would reach a piece so tiny that it could no longer be divided. Democritus called these infinitesimally small pieces of matter atomos, meaning 'indivisible'. He suggested that atomos were eternal and could not be destroyed. Democritus theorized that atomos were specific to the material that they made up, meaning that the atomos of stone were unique to stone and different from the atomos of other materials, such as fur. This was a remarkable theory that attempted to explain the whole physical world in terms of a small number of ideas.
Ultimately, though, Aristotle and Plato, two of the best-known philosophers of Ancient Greece, rejected the theories of Democritus. Aristotle accepted the theory of Empedocles, adding his own (incorrect) idea that the four core elements could be transformed into one another. Because of Aristotle's great influence, Democritus's theory would have to wait almost 2,000 years before being rediscovered.
In the 17th and 18th centuries CE, several key events helped revive the theory that matter was made of small, indivisible particles. In 1643, Evangelista Torricelli, an Italian mathematician and pupil of Galileo, showed that air had weight and was capable of pushing down on a column of liquid mercury (thus inventing the barometer). This was a startling finding. If air – this substance that we could not see, feel, or smell – had weight, it must be made of something physical. But how could something have a physical presence, yet not respond to human touch or sight? Daniel Bernoulli, a Swiss mathematician, proposed an answer. He developed a theory that air and other gases consist of tiny particles that are too small to be seen, and are loosely packed in an empty volume of space. The particles could not be felt because unlike a solid stone wall that does not move, the tiny particles move aside when a human hand or body moves through them. Bernoulli reasoned that if these particles were not in constant motion, they would settle to the ground like dust particles; therefore, he pictured air and other gases as loose collections of tiny billiard-ball-like particles that are continuously moving around and bouncing off one another.
Law of Conservation of Mass

Many scientists were busy studying the natural world at this time. Shortly after Bernoulli proposed his theory, the Englishman Joseph Priestley began to experiment with red mercury calx in 1773. Mercury calx, a red solid stone, had been known and coveted for thousands of years because when it is heated, it appears to turn into mercury, a silver liquid metal. Priestley had observed that it does not just turn into mercury; it actually breaks down into two substances when it is heated, liquid mercury and a strange gas. Priestley carefully collected this gas in glass jars and studied it. After many long days and nights in the laboratory, Priestley said of the strange gas, "What surprised me more than I can well express was that a candle burned in this air with a remarkably vigorous flame." Not only did flames burn strongly in this gas, but a mouse placed in a sealed container of this gas lived for a longer period of time than a mouse placed in a sealed container of ordinary air. Priestley's discovery revealed that substances could combine together or break apart to form new substances with different properties. For example, a colorless, odorless gas could combine with mercury, a silver metal, to form mercury calx, a red mineral.
Priestley called the gas he discovered dephlogisticated air, but this name would not stick. In 1778, Antoine Lavoisier, a French scientist, conducted many experiments with dephlogisticated air and theorized that the gas made some substances acidic. He renamed Priestley's gas oxygen, from the Greek words that loosely translate as 'acid maker'. While Lavoisier's theory about oxygen and acids proved incorrect, his name stuck. Lavoisier knew from other scientists before him that acids react with some metals to release another strange and highly flammable gas called phlogiston. Lavoisier mixed the two gases, phlogiston and the newly renamed oxygen, in a closed glass container and inserted a match. He saw that phlogiston immediately burned in the presence of oxygen, and afterwards he observed droplets of water on the glass container. After careful testing, Lavoisier realized that the water was formed by the reaction of phlogiston and oxygen, and so he renamed phlogiston hydrogen, from the Greek words for 'water maker'.
Lavoisier also burned other substances such as phosphorus and sulfur in air, and showed that they combined with air to make new materials. These new materials weighed more than the original substances, and Lavoisier showed that the weight gained by the new materials was lost from the air in which the substances were burned. From these observations, Lavoisier established the Law of Conservation of Mass, which says that mass is not lost or gained during a chemical reaction.
Comprehension Checkpoint
Modern atomic theory

Priestley, Lavoisier, and others had laid the foundations of the field of chemistry. Their experiments showed that some substances could combine with others to form new materials, other substances could be broken apart to form simpler ones, and a few key "elements" could not be broken down any further. But what could explain this complex set of observations? John Dalton, an exceptional British teacher and scientist, put together the pieces and developed the first modern atomic theory in 1803. To learn more about Priestley's and Lavoisier's experiments and how they formed the basis of Dalton's theories, try the interactive experiment Dalton's Playhouse, linked to below.

Interactive Animation:
Dalton's Playhouse
Dalton made it a regular habit to track and record the weather in his hometown of Manchester, England. Through his observations of morning fog and other weather patterns, Dalton realized that water could exist as a gas that mixed with air and occupied the same space as air. Solids could not occupy the same space as each other; for example, ice could not mix with air. So what could allow water to sometimes behave as a solid and sometimes as a gas? Dalton realized that all matter must be composed of tiny particles. In the gas state, those particles floated freely around and could mix with other gases, as Bernoulli had proposed. But Dalton extended this idea to apply to all matter – gases, solids, and liquids. Dalton first proposed part of his atomic theory in 1803 and later refined these concepts in his classic 1808 paper A New System of Chemical Philosophy (which you can access through a link under the Resources tab).
Dalton's theory had four main concepts:
-
All matter is composed of indivisible particles called atoms. Bernoulli, Dalton, and others pictured atoms as tiny billiard-ball-like particles in various states of motion. While this concept is useful to help us understand atoms, it is not correct as we will see in later modules on atomic theory linked to at the bottom of this module.
-
All atoms of a given element are identical; atoms of different elements have different properties. Dalton's theory suggested that every single atom of an element such as oxygen is identical to every other oxygen atom; furthermore, atoms of different elements, such as oxygen and mercury, are different from each other. Dalton characterized elements according to their atomic weight; however, when isotopes of elements were discovered in the late 1800s, this concept changed.
-
Chemical reactions involve the combination of atoms, not the destruction of atoms. Atoms are indestructible and unchangeable, so compounds, such as water and mercury calx, are formed when one atom chemically combines with other atoms. This was an extremely advanced concept for its time; while Dalton's theory implied that atoms bonded together, it would be more than 100 years before scientists began to explain the concept of chemical bonding.
-
When elements react to form compounds, they react in defined, whole-number ratios. The experiments that Dalton and others performed showed that reactions are not random events; they proceed according to precise and well-defined formulas. This important concept in chemistry is discussed in more detail below.
Comprehension Checkpoint
Some of the details of Dalton's atomic theory require more explanation.
Elements: As early as 1660, Robert Boyle recognized that the Greek definition of element (earth, fire, air, and water) was not correct. Boyle proposed a new definition of an element as a fundamental substance, and we now define elements as fundamental substances that cannot be broken down further by chemical means. Elements are the building blocks of the universe. They are pure substances that form the basis of all of the materials around us. Some elements can be seen in pure form, such as mercury in a thermometer; some we see mainly in chemical combination with others, such as oxygen and hydrogen in water. We now know of approximately 116 different elements. Each of the elements is given a name and a one- or two-letter abbreviation. Often this abbreviation is simply the first letter of the element; for example, hydrogen is abbreviated as H, and oxygen as O. Sometimes an element is given a two-letter abbreviation; for example, helium is He. When writing the abbreviation for an element, the first letter is always capitalized and the second letter (if there is one) is always lowercase.
Atoms: A single unit of an element is called an atom. The atom is the most basic unit of matter, which makes up everything in the world around us. Each atom retains all of the chemical and physical properties of its parent element. At the end of the nineteenth century, scientists would show that atoms were actually made up of smaller, "subatomic" pieces, which smashed the billiard-ball concept of the atom (see our Atomic Theory I: The Early Days module).
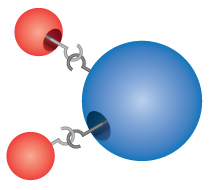
Compounds: Most of the materials we come into contact with are compounds, substances formed by the chemical combination of two or more atoms of the elements. A single "particle" of a compound is called a molecule. Dalton incorrectly imagined that atoms "hooked" together to form molecules. However, Dalton correctly realized that compounds have precise formulas. Water, for example, is always made up of two parts hydrogen and one part oxygen. The chemical formula of a compound is written by listing the symbols of the elements together, without any spaces between them. If a molecule contains more than one atom of an element, a number is subscripted after the symbol to show the number of atoms of that element in the molecule. Thus the formula for water is H2O, never HO or H2O2.
Comprehension Checkpoint
Law of Definite Proportions
The idea that compounds have defined chemical formulas was first proposed in the late 1700s by the French chemist Joseph Proust. Proust performed a number of experiments and observed that no matter how he caused different elements to react with oxygen, they always reacted in defined proportions. For example, two parts of hydrogen always reacts with one part oxygen when forming water; one part mercury always reacts with one part oxygen when forming mercury calx. Dalton used Proust's Law of Definite Proportions in developing his atomic theory.
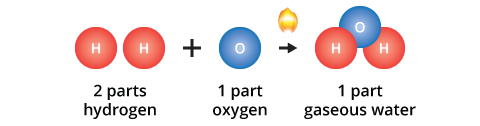
The law also applies to multiples of the fundamental proportion, for example:
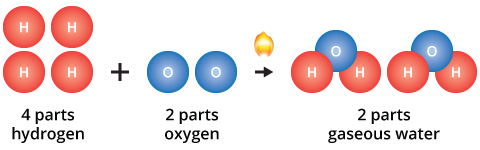
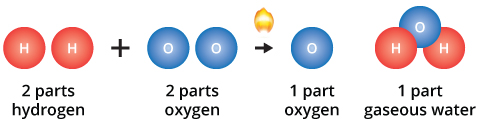
In both of these examples, the ratio of hydrogen to oxygen to water is 2 to 1 to 1. When reactants are present in excess of the fundamental proportions, some reactants will remain unchanged after the chemical reaction has occurred.
The story of the development of modern atomic theory is one in which scientists built upon the work of others to produce a more accurate explanation of the world around them. This process is common in science, and even incorrect theories can contribute to important scientific discoveries. Dalton, Priestley, and others laid the foundation of atomic theory, and many of their hypotheses are still useful. However, in the decades after their work, other scientists would show that atoms are not solid billiard balls, but complex systems of particles. Thus, they would smash apart a bit of Dalton's atomic theory in an effort to build a more complete view of the world around us.
Table of Contents
Activate glossary term highlighting to easily identify key terms within the module. Once highlighted, you can click on these terms to view their definitions.
Activate NGSS annotations to easily identify NGSS standards within the module. Once highlighted, you can click on them to view these standards.